In Part 1, we introduced the Frailty Paradox: the NHS is already delivering many of the components of effective frailty care, but these are not connected into a coordinated system.
We also set out population health management as the model the system is trying to achieve. This depends on the ability to identify need, prioritise risk, deliver appropriate intervention, and monitor outcomes across a defined population.
In Part 2, we explored the first of these capabilities: frailty identification, which must be accurate, scalable across the whole population, enable early identification, and be simple for clinicians to use.
Existing tools only partially meet these requirements, with population-level approaches lacking clinical precision and clinical approaches lacking systematic reach.
This article sets out how frailty identification can become systematic, clinical, and continuous.
The Pathfields Tool: Connecting Identification and Diagnosis
The Pathfields Tool was developed as a practical response to this problem, combining population-level identification with clinician-led diagnosis(1).
It was applied across the registered population aged 65 years and over. Within this population, individuals were identified as being at higher risk of undiagnosed frailty if they had one or more of the following characteristics:
• Age 90 years or over
• Diagnosis of dementia
• Residence in a care home
• Housebound status or a recent home visit
• Observed mobility problems during consultations
Observed mobility problems were particularly important. A central design principle was the systematic identification of mild frailty. Unlike moderate and severe frailty, this group was largely ambulatory and continued to attend the surgery for routine care such as blood pressure checks, flu vaccination, and nurse-led clinics, providing a consistent point of contact within everyday clinical practice. This was used deliberately to create an identification pathway: saving a consultation record prompted nursing staff to code observed mobility problems, generating an early signal that brought patients into the higher-risk cohort for subsequent clinical assessment.
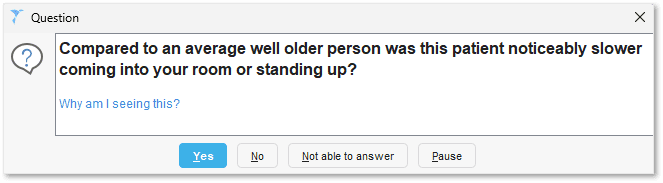
Figure 1: the nurse or HCA prompt used to capture mobility issues during routine consultations.
When a patient in one of these groups consulted, saving their record triggered a prompt to code frailty status using four categories:
• Not frail
• Mild frailty
• Moderate frailty
• Severe frailty
Assessment was supported by pictorial representations from the Rockwood Clinical Frailty Scale and informed by the clinician’s longitudinal knowledge of the patient.

Figure 2. Clinician popup (Clinical Frailty Scale diagrams reproduced with permission from Dalhousie University).
After coding frailty, clinicians recorded place of residence (for example nursing home, residential home, supported living, or at home), with those at home further classified as housebound or not. This allowed the population to be segmented first by setting and then, within the home-based cohort, by functional status.
The tool also flagged patients on the learning disability register or with stable neurological conditions before the prompt appeared, reducing the risk of misclassification where pictorial representations may be misleading.
Once completed, the tool became dormant for 12 months, ensuring the process was time-neutral and avoiding repeated prompts. Because frailty was clinician-diagnosed and recorded, it enabled longitudinal tracking, supporting population health management and giving insight into frailty progression(1).
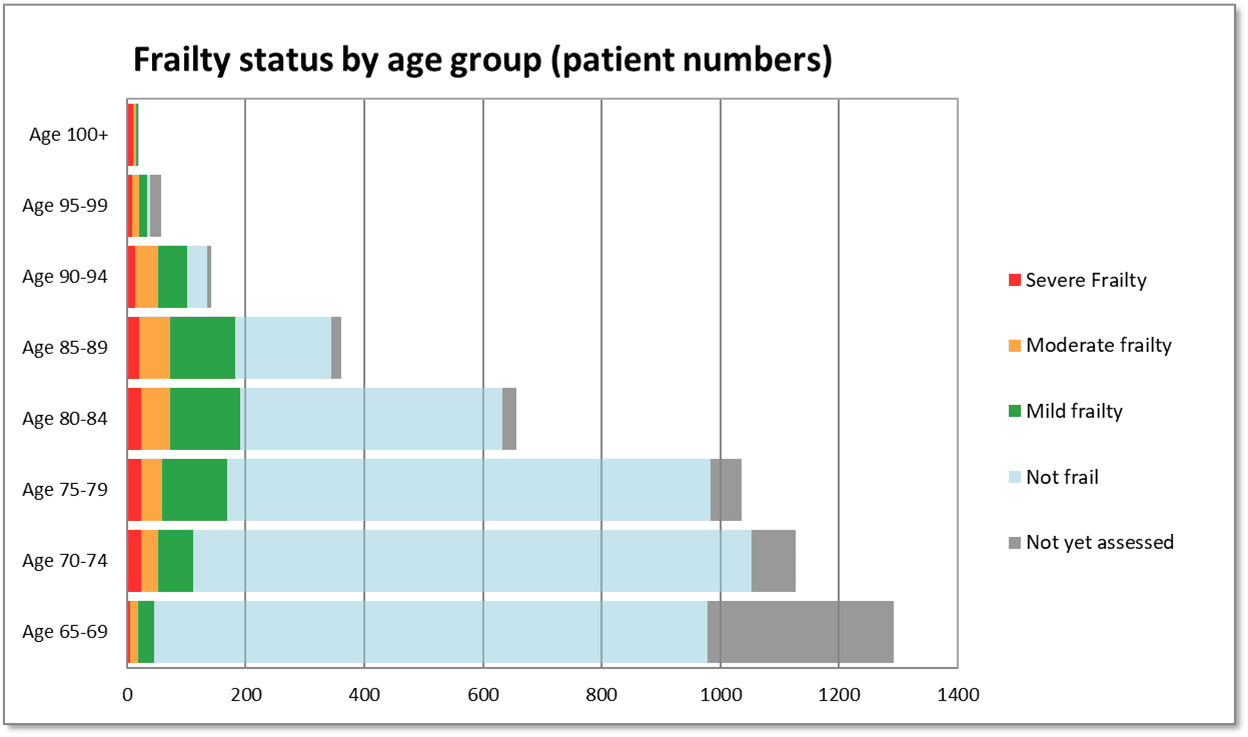
Figure 3. Frailty prevalence by age group in a single general practice population, identified using the Pathfields Tool (n=30,000).
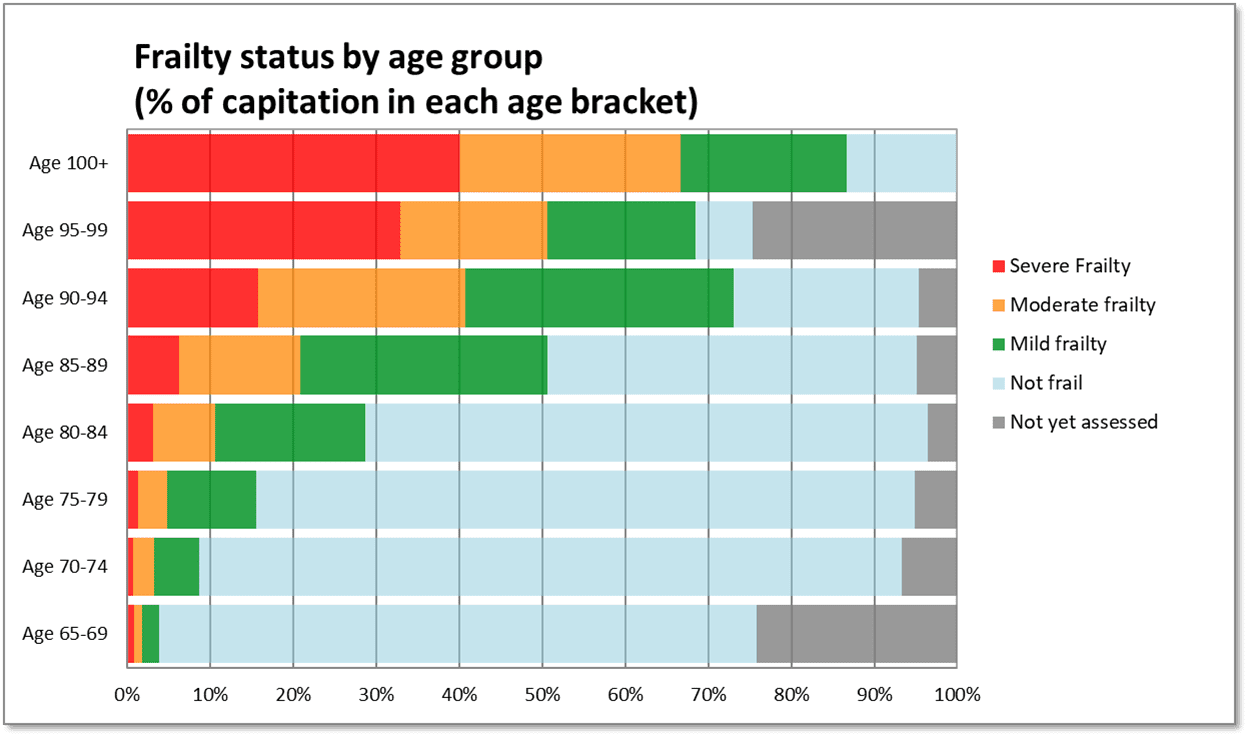
Figure 4. Frailty prevalence (%) by age group in a single general practice population, identified using the Pathfields Tool (n=30,000).
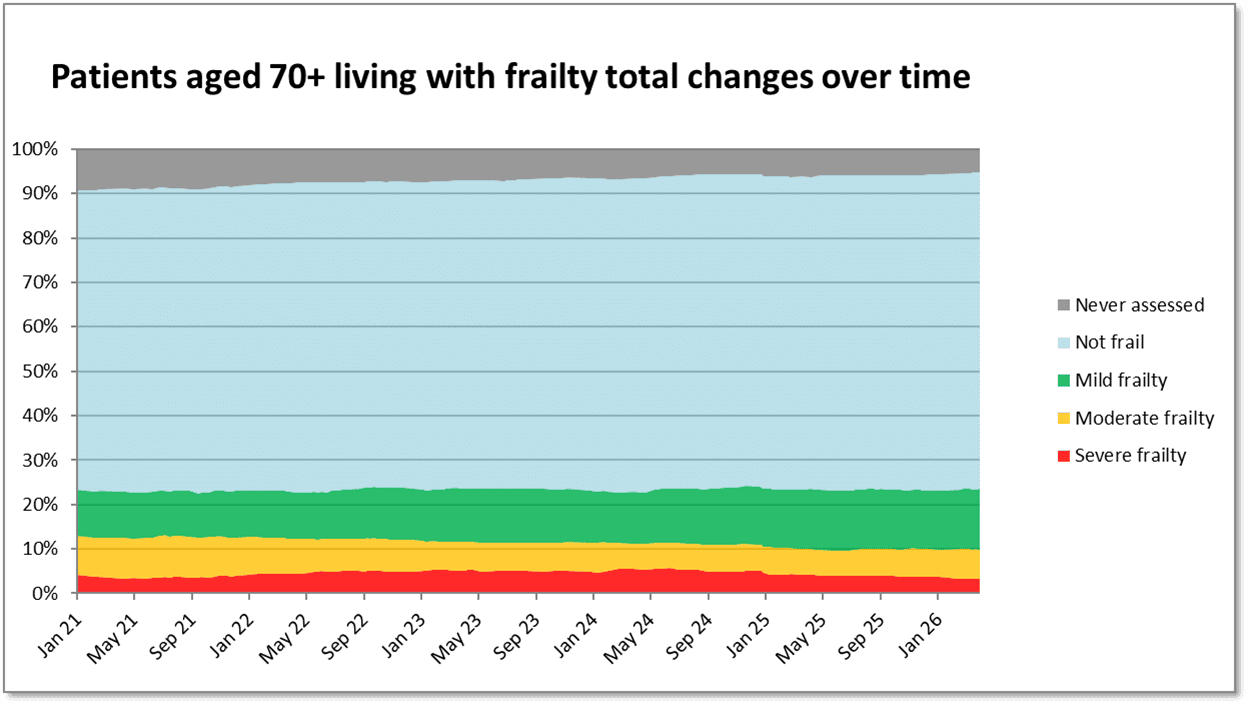
Figure 5. Longitudinal changes in frailty status over 6 years in patients aged 70+ years within a single general practice population, identified using the Pathfields Tool (n=30,000).
In effect, the Pathfields Tool created a continuously self-replenishing frailty register, shifting frailty identification from a static, list-based exercise to a dynamic process embedded within routine care. Patients flowed into the cohort through everyday clinical contact, with frailty captured by severity and setting, creating a continuously updated population view of frailty.
What the Early Data Showed
Early data from a single general practice (n=4,552 patients aged 65 and over) suggested improved identification.
The Pathfields Tool identified 1,348 patients within the high-risk cohort, of whom 951 (70.5%) were clinically assessed:
- 24.1% not frail
- 75.9% frail (27.3% mild; 29.9% moderate; 18.7% severe)
By comparison, the eFI identified 683 patients, of whom 598 (87.6%) were assessed:
- 52.7% not frail
- 47.3% frail (19.1% mild; 17.2% moderate; 11.2% severe)
Extrapolated across the population, this suggested a frailty prevalence of 22.5% (1,024/4,552) in those aged 65 and over(1).
In practical terms, this meant that for every 10 patients identified as living with frailty using the Pathfields Tool, around 6 were not identified by the eFI. While derived from a single population, this highlights how different approaches can identify different groups of patients.
Clinician feedback was positive. The use of a single-click prompt, supported by pictorial guidance, enabled rapid categorisation without adding time to consultations. In contrast, the eFI required dedicated clinician time to review patient lists and manually confirm frailty through notes review.
From Identification to Action: Use During the Pandemic
During the COVID-19 pandemic, the Pathfields Tool supported proactive population health management within a Primary Care Network serving 30,000 patients(2).
It enabled identification of patients with frailty who were not included on national shielding lists, allowing targeted intervention:
- Severe frailty: contacted and offered advance care planning
- Moderate frailty: supported by social prescribing teams, ensuring access to food, medication, and support
- Mild frailty: provided with health and wellbeing information to prevent deconditioningPatients with severe frailty were contacted and offered advance care planning
This demonstrated how systematic identification could support coordinated, proactive care at neighbourhood level(2).
Limitations in the Evidence
There are several limitations.
First, the tool is currently implemented within SystmOne and not yet available in EMIS, which may limit adoption in some settings.
Second, inter-observer reliability was not formally assessed. However, observed patterns suggested internal consistency. The distribution of frailty severity aligned with expected patterns of dependency and residence: patients with mild frailty were predominantly not housebound, while those with moderate and severe frailty were more likely to be housebound or living in care homes. This alignment with established frailty descriptors suggests that increased identification reflects improved detection rather than systematic over-classification(1).
Most importantly, the published evidence is derived from a single general practice, and further evaluation across diverse populations and settings is required to establish generalisability. The findings do, however, reflect routine use across multiple clinicians in everyday practice, providing an early indication of real-world feasibility(1).
From Evidence to Adoption
Since publication, use in practice has already expanded beyond the original evaluation site. The Pathfields Tool is now in routine use across a growing number of organisations, including 38 general practices, an SDEC service within an acute trust, and a community services provider, reflecting its ability to operate within existing resources and clinical practice.
The Pathfields Tool is freely available for use across NHS organisations. Further information, including how to access it, is available here:
From Case Finding to Continous Frailty Identification
The significance of the Pathfields Tool is not simply that it identifies frailty, but that it transforms identification from a one-off task into a continuous process. By embedding detection within everyday clinical practice, it creates a self-replenishing frailty register that is constantly updated through routine care. This represents a practical way of addressing the Frailty Paradox: not through new activity, but by allowing a continuously updated, population-level view of frailty to emerge.
Where this series goes next
Identifying frailty is only the starting point.
Once a population becomes visible, not everyone can be seen at once. The question becomes who is most at risk, and who needs to be reached first.
In the next article, we will explore how signals of deterioration are used to prioritise risk across a neighbourhood population, and how that determines who is seen in time to change outcomes.
References
- Attwood D, et al. The Pathfields Tool: a frailty case-finding tool using primary care IT—implications for population health management. Age Ageing. 2020;49(6):1087–1092. doi:10.1093/ageing/afaa119
- Capturing Beneficial innovations from the COVID-19 pandemic BGS. 2021.