Prioritising risk in frailty across neighbourhood populations
In Part 1, we set out the four capabilities required for population health management of frailty across a neighbourhood population: identify, prioritise, intervene, and monitor. Parts 2 and 3 focused on identification, its challenges, and solutions. This article moves to the next step: prioritisation.
This is where the Frailty Paradox deepens.
The NHS understands frailty. It measures admissions, ambulance conveyances, and crisis in detail. It also understands many of the factors that lead to deterioration.
Yet in practice, the system remains focused on the consequences of decline, rather than the signals that predict it.
The signals are there.
The question is whether the system uses them to decide who it prioritises, today.
Prioritisation is a matter of life and death
Prioritisation in frailty is where outcomes are decided. As frailty increases, survival falls. Many people living with advanced frailty die within relatively short timeframes, often during a hospital admission or shortly afterwards (1,2,3,4).
This creates a simple but uncomfortable reality: You do not have time to get to everyone.
Across any neighbourhood population, some people with frailty will deteriorate, and some will die before the system reaches them.
But this is not fixed.
When early signals of deterioration are recognised and acted on, outcomes change. Deterioration can be slowed, stabilised, and in some cases reversed. Admissions can be avoided. Independence can be maintained for longer.
This is where prioritisation becomes critical.
High-risk markers do not occur in isolation, they reveal unmet need
The system is not short of signals predicting deterioration. The challenge is recognising that, in frailty, they rarely occur in isolation and are not discrete problems to solve, but highly visible manifestations of deeper, interconnected unmet need.
Take a fall. On the surface, it appears to be a single event.
In frailty, it is a signal event, the point at which multiple pressures become visible at the same time:
- Biological: muscle weakness, poorly controlled long term conditions, medication effects
- Functional: reduced mobility, impaired balance, difficulty with activities of daily living
- Psychological: low mood, fear of falling, cognitive impairment
- Social: isolation, limited support, unsafe environment, carer strain
The fall is not the cause. It is the moment the system notices.
If it is treated as an isolated issue, the response remains reactive.
If it is recognised as a proxy for wider instability, the response becomes proactive.
That distinction determines whether care stabilises the person or simply manages the consequence.
Signals are everywhere, but not yet connected
The same pattern applies across multiple markers of risk:
- Falls, fractures, and declining mobility
- Escalating healthcare utilisation (ambulance, emergency department, hospital, and primary care)
- Malnutrition
- High-risk medications
- Delirium
- Clinician concern
- Carer burnout
In frailty, these are not just signals of deterioration, but indicators of underlying unmet biological, functional, psychological, and social need.
They may occur alone, or alongside other signals. Either way, they are rarely random. They are points at which need becomes visible.
A missed signal: HbA1c as a marker of instability
Some of the most powerful signals are not always recognised as such.
HbA1c is a good example.
In people living with frailty, risk exists at both ends of the spectrum:
- An HbA1c above 75 mmol/mol is associated with infection (5,6,7) and osmotic symptoms (8), which can precipitate falls and delirium, leading to hospitalisation, loss of independence, and mortality (9,10).
- Conversely, in those treated with insulin or sulphonylureas, an HbA1c below 48 mmol/mol increases the risk of hypoglycaemia, with similarly harmful consequences (11).
Maintaining glycaemic control requires a continuous series of daily decisions (12). When those decisions are not made, control deteriorates.
In frailty, poor glycaemic control is more than a clinical problem. It is a powerful signal that other biological, functional, psychological, or social needs are not being met, and have taken precedence (13).
This makes HbA1c a clear marker that something wider is failing.
From fragmented signals to visible risk
Signals of deterioration are routinely captured across the system but held in isolation within the service that generated them. As we observed in Part 1, “the activity exists, but the system to connect it does not.”
As a result, prioritisation fails.
Taking the example of a fall again, a fall resulting in an Emergency Department attendance may be managed as a single event in isolation. But when that same event is seen in general practice, alongside prior falls and inclusion on a dynamic frailty register, it becomes something different entirely: part of a pattern that signals underlying instability.
This is the shift that allows risk to become visible, enabling earlier, coordinated action.
The next question is how this visibility can be brought together in practice.
General practice: where signals already converge
General practice is the logical anchor point. It already holds the closest thing the NHS has to a single source of truth: a longitudinal record, system-wide correspondence, and a continuously updated frailty register.
Signals converge here.
Yet the NHS has not organised itself around general practice to act on them.
What this looks like in practice: from signals to prioritised cohorts
The question, then, is what this looks like when applied in practice.
By combining frailty identification using the Pathfields Tool with key signals described above, targeted searches create priority cohorts, patients at high risk who require coordinated action.
Three examples illustrate this, and how different signals drive different types of response.
1. High-risk medication signals: Triage and targeted review
Aligning the frailty register with high-risk prescribing identifies patients where medication may be contributing to deterioration, but more importantly, signals the presence of wider unmet need.
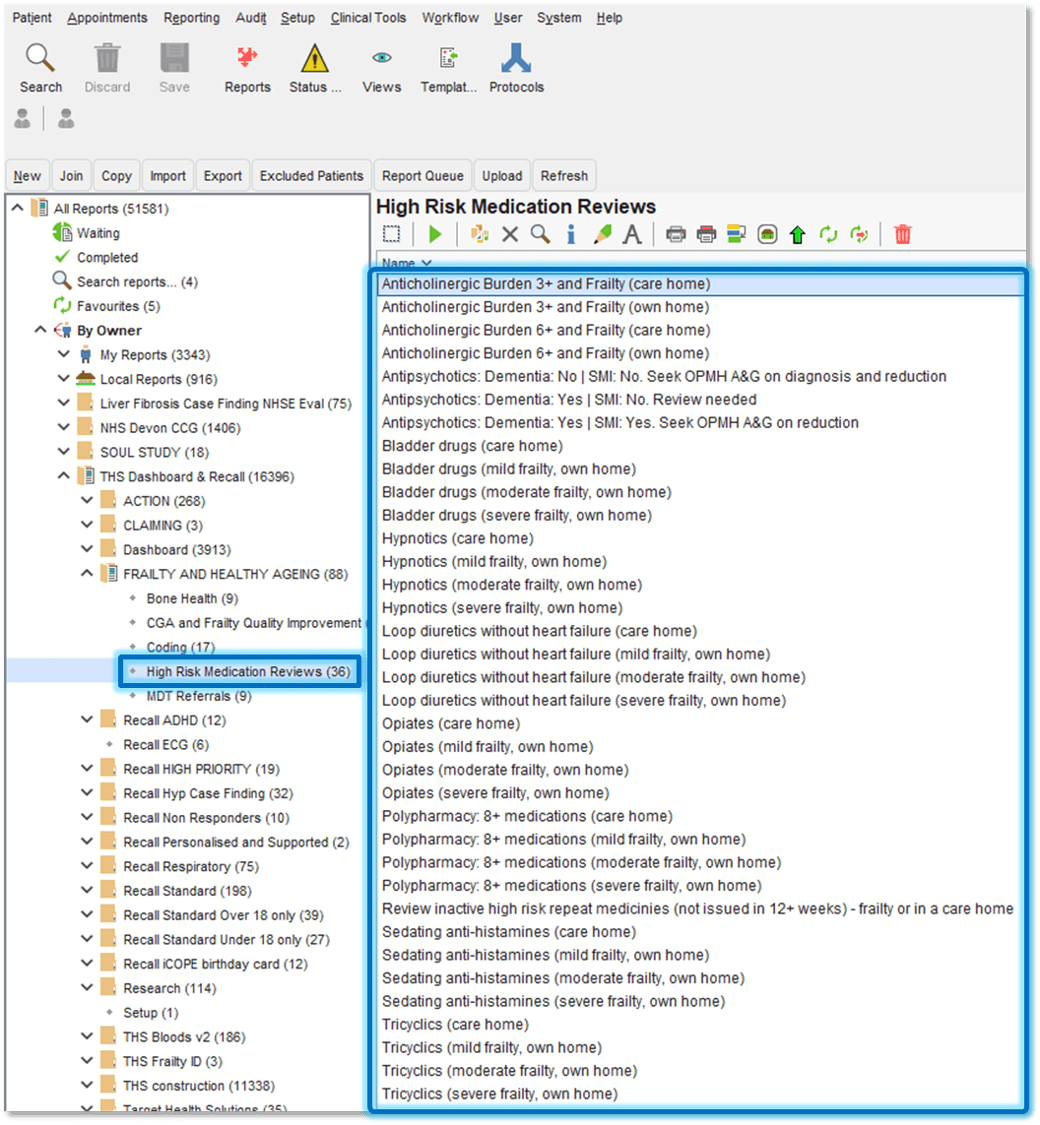
Figure 1: High-risk prescribing, when used as a signal and combined with frailty status, surfaces patients where medication may both contribute to instability and signal underlying unmet need.
From here, specific searches can be selected. For example, choosing tricyclic antidepressants generates a list of patients living with frailty exposed to that risk.
At this point, the signal becomes a point of triage. Where it represents a single issue, this may lead to a targeted uniprofessional intervention, for example a medication review by a pharmacist. Where multiple issues are present, it prompts escalation to MDT review.
Opening an individual patient in iCGA 3.0 brings these signals together, allowing the clinician to see whether this is an isolated issue or part of a broader pattern of unmet need contributing to overall risk. In practice, this often reveals multiple overlapping concerns. In this example, a tricyclic prescription sits alongside other high-risk medications, unmet bone health needs, sub-optimally controlled long-term conditions, and recent healthcare use, indicating a patient who may benefit from MDT discussion.
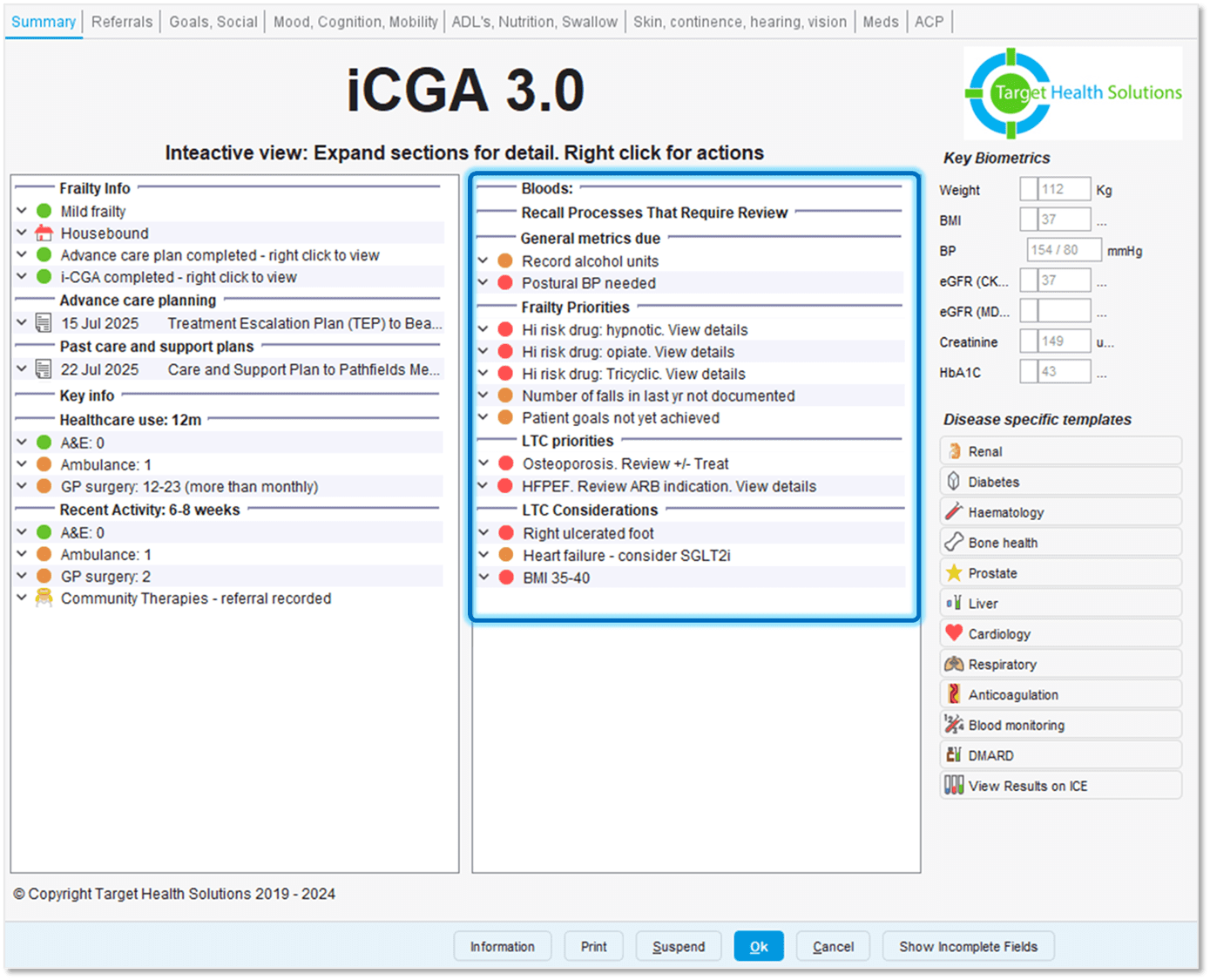
Figure 2: Individual patient view (iCGA 3.0) showing how a single prescribing signal reveals multiple areas of unmet need across biological, functional, and social domains.
This illustrates how a tricyclic prescription, when seen not just as an isolated event but as a signal, reveals a broader pattern of unmet need and risk.
2. High-risk signals of instability: Direct to MDT Review
The same principle applies to other high-risk signals such as recurrent falls, hospital admissions, malnutrition, and fractures. When seen not as isolated events but as signals, they reveal broader patterns of unmet need.
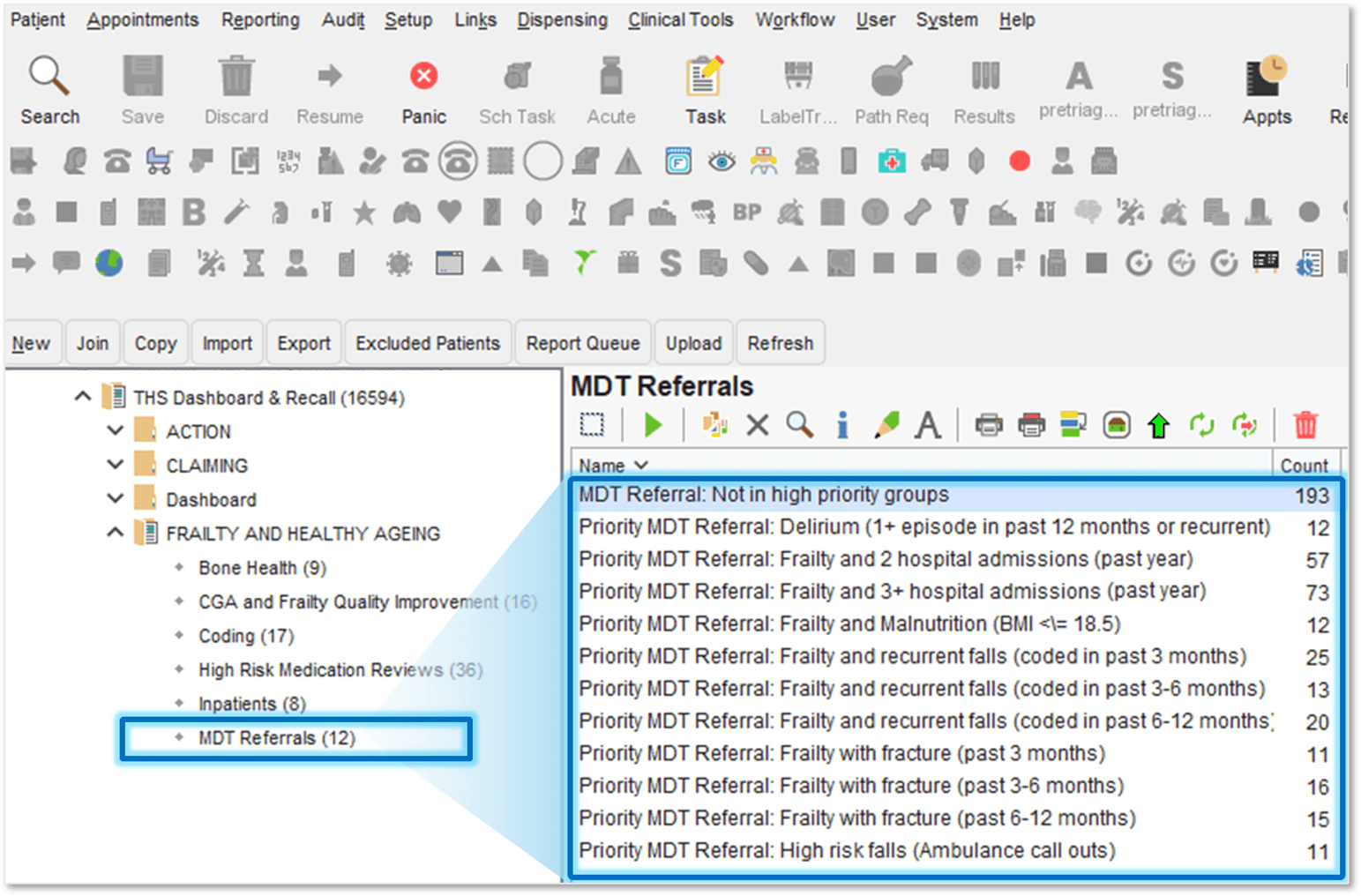
Figure 3: Targeted MDT search combining frailty with high-risk signals such as falls, admissions, malnutrition, and fracture to identify patients requiring coordinated intervention.
Here, the distinction is important.
Unlike medication signals, which often act as a point of triage, these signals represent established instability and a changing clinical trajectory.
As a result, the response is not triage but escalation. These patients are likely to require coordinated multidisciplinary review to address the underlying biological, functional, psychological, and social drivers of deterioration.
3. Inpatients: acting in real time
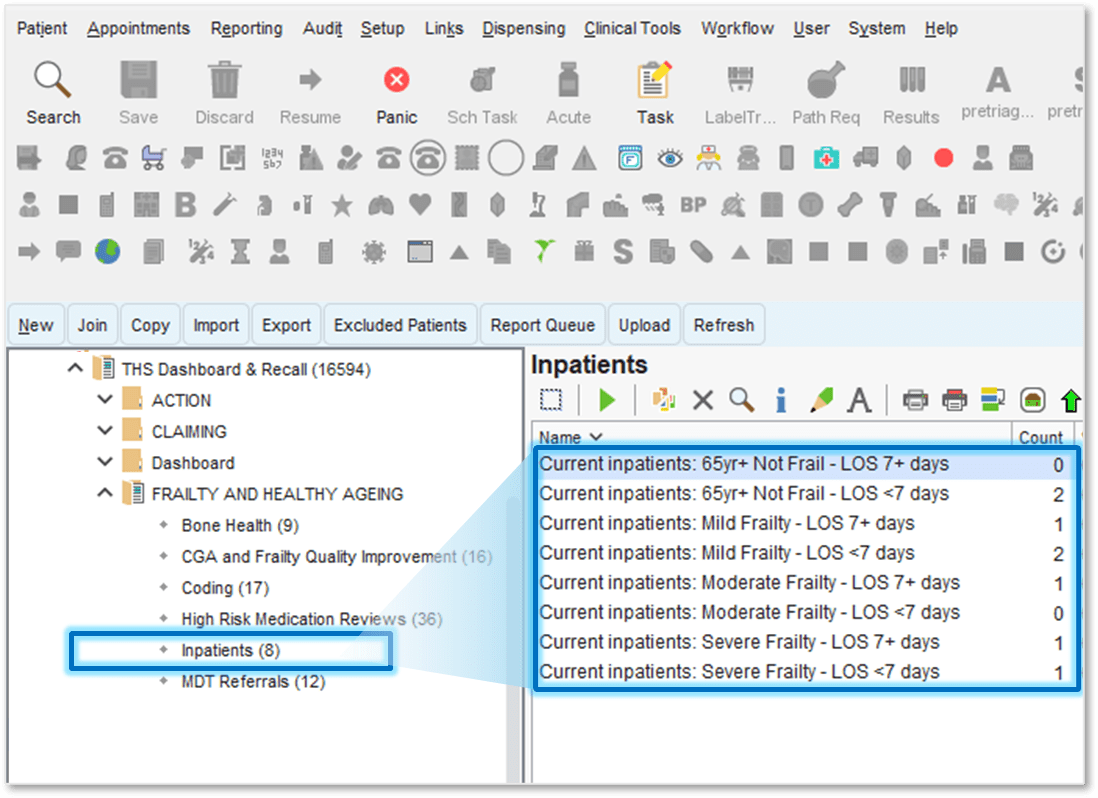
Figure 4: Inpatient search stratified by frailty and length of stay, identifying patients at risk of deconditioning and delayed discharge for early neighbourhood team input.
For patients already in hospital, inpatient searches provide real-time visibility. Stratifying current admissions by frailty and length of stay highlights those at risk of deconditioning and delayed discharge.
Used as a signal, this enables earlier, coordinated action, with community and neighbourhood teams working alongside hospital teams during the admission to support a safe return home, whilst addressing unmet need identified through the CGA process.
From Signals to Systematic Prioritisation
Across each of these examples, the principle is the same.
Frailty identification provides the population.
Signals provide the context.
Together, they define who should be prioritised.
When general practice is used as the anchor point, these signals converge through continuity, longitudinal knowledge, and a single source of truth.
Prioritisation becomes systematic, transparent, and shared, no longer dependent on individual clinicians recognising patterns, but embedded within the system itself.
Where this series goes next
Prioritisation only has value if the system can respond to it.
Action must be coordinated, continuous, and address the unmet need these signals reveal.
The question now is how the system organises itself to deliver that, consistently, at neighbourhood level.
References
- Attwood D, et al. Does proactive care in care homes improve survival? A quality improvement project. BMJ Open Qual. 2024 Jun 4;13(2):e002771. doi: 10.1136/bmjoq-2024-002771. PMID: 38834371; PMCID: PMC11163642.
- Javadzadeh D, et al. Clinical Frailty Scale score is a predictor of short-, mid- and long-term mortality in critically ill older adults (≥ 70 years) admitted to the emergency department: an observational study. BMC Geriatr. 2024 Oct 21;24(1):852. doi: 10.1186/s12877-024-05463-7. PMID: 39434029; PMCID: PMC11492669.
- Munir, Ehrlington et al. Frailty is associated with 30-day mortality: a multicentre study of Swedish emergency departments. Emergency Medicine Journal 2024;41:514-519. doi:10.1136/emermed-2023-213444
- BlytheR, et al. Validation of the Clinical Frailty Scale for predicting 90-day mortality in hospitalised older adults screened as at risk of nearing the end of life in Queensland, Australia: a multisite observational study. BMJ Open 2025;15: doi: 10.1136/bmjopen-2025-1084193. 3. Peng Y, et al. Frailty and risks of all-cause and cause-specific death in community-dwelling adults: a systematic review and meta-analysis. BMC Geriatr 2022;22:725. doi:10.1186/s12877-022-03404-w
- Zacay, et al. Glycemic Control and Risk of Cellulitis. Diabetes Care1 February 2021; 44 (2): 367–372. https://doi.org/10.2337/dc19-1393
- Carey I, et al. Effects of long-term HbA1c variability on serious infection risks in patients with type 2 diabetes and the influence of age, sex and ethnicity: A cohort study of primary care data. Diabetes Res Clin Pract. 2024 May;211:111641. doi: 10.1016/j.diabres.2024.111641. Epub 2024 Mar 26. PMID: 38548108.
- Reduced risk of infection with well-controlled HBA1c levels. European Medical Journal. 2018
- Seker et al. Orthostatic hypotension in type 2 diabetes: Prevalance and risk factors. Medicina Clinica 2026 166(1) 107281, ISSN 0025-7753, https://doi.org/10.1016/j.medcli.2025.107281.
- Gale CR, et al. Prevalence and risk factors for falls in older men and women: The English Longitudinal Study of Ageing. Age Ageing. 2016 Nov;45(6):789-794. doi: 10.1093/ageing/afw129. Epub 2016 Jul 19. PMID: 27496938; PMCID: PMC5105823.
- Happanen, M. et al. Delirium and adverse clinical outcomes: a matched cohort study in the UK Biobank. The Lancet Healthy Longevity, 2026 7(1) 100816, ISSN 2666-7568, https://doi.org/10.1016/j.lanhl.2025.100816.
- Fløde M, et al. Hypoglycaemia and its associations with diabetes and age-related factors in older home-dwelling people with diabetes. BMC Geriatr. 2025 25(1):1020. doi: 10.1186/s12877-025-06732-9. PMID: 41398947; PMCID: PMC12706898.
- Easing the Burden for People with Diabetes. Diabetes UK. 2022 (web address: https://www.diabetes.org.uk/about-us/news-and-views/easing-burden-people-diabetes)
- Howell C, et al. Perspective: Acknowledging a Hierarchy of Social Needs in Diabetes Clinical Care and Prevention. Diabetes Metab Syndr Obes. 2023 19(16):161-166. doi: 10.2147/DMSO.S389182. PMID: 36760578; PMCID: PMC9869784.

