In Part 1,we set out the Frailty Paradox: the NHS already delivers many components of effective frailty care, but not as a coordinated system.
In Parts 2 and 3 we examined the limitations of current approaches to frailty identification, and introduced the Pathfields Tool as a more accurate, scalable, and clinically usable method.
In Part 4, we explored how signals of deterioration can be combined with frailty identification to prioritise risk across a population, making visible who needs to be seen first.
But prioritisation only has value if the system can respond to it.
Population health management is defined not by how well a system identifies and prioritises need, but by what it does next.
Population Health Management in Frailty
Population health management in frailty involves:
- Systematically identifying people across the full spectrum of frailty, including those within high-risk cohorts
- Grouping them into meaningful categories such as fit, mild, moderate, and severe frailty
- Delivering coordinated, evidence-based interventions at scale
This model is well understood, but in practice remains fragmented, with care delivered in isolation rather than as a coordinated pathway across the full lifecourse.
The population pyramid below describes a layered model of care, where the type of intervention changes as frailty progresses.
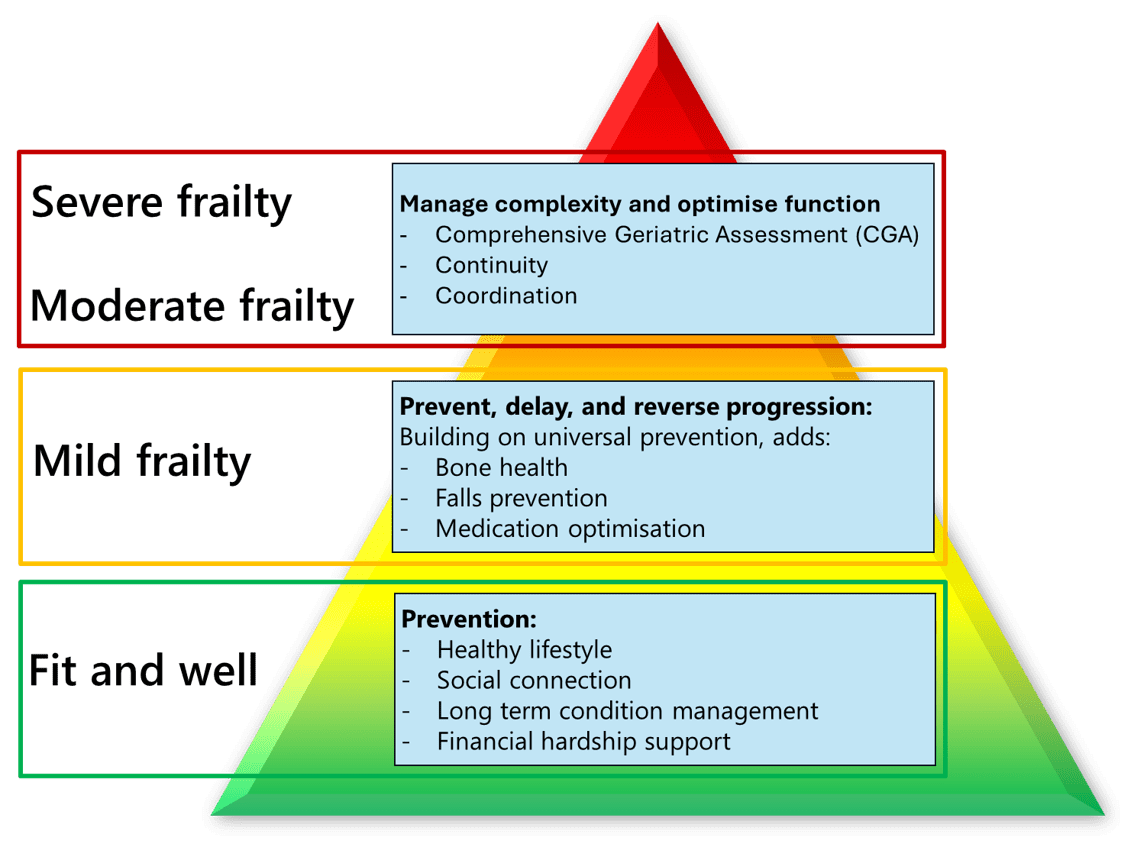
Figure 1: A population health approach to frailty: identify and segment the population, then offer personalised, evidence based interventions. For moderate to severe frailty, this requires continuous, coordinated CGA.
At the base, for people who are fit and well, the focus is on universal prevention:
- Supporting a healthy lifestyle, including physical activity and good nutrition
- Maintaining social connection and reducing isolation
- Optimising long-term condition management
- Identifying and supporting financial hardship
As people move into mild frailty, the focus shifts to preventing and, where possible, reversing progression by maintaining function and independence:
- Building on universal prevention
- Falls prevention
- Bone health
- Medication optimisation, particularly reducing high risk medications
At this level, there is a real opportunity to change trajectory and delay or even reverse decline.
Further up the pyramid, in moderate and severe frailty, the model changes more fundamentally.
Here, the challenge is no longer simply identifying risk or adding isolated interventions.
It is managing complexity through a coordinated, continuous, Comprehensive Geriatric Assessment (CGA).
What is a Comprehensive Geriatric Assessment (CGA)?
CGA is a holistic process that brings together medical, functional, psychological, and social aspects of care. It is delivered by a multidisciplinary team over multiple interactions, rather than by a single professional.
Following assessment, a personalised care and support plan is co-produced with the individual, involving those important to them, such as family, friends, or carers, where appropriate.
CGA must be delivered across both proactive and urgent care settings. The difference is the pace of delivery.
What does the evidence for CGA show?
In acute settings, CGA has been shown to improve outcomes, including increasing the likelihood that people are alive and living in their own homes, alongside improvements in function, readmission, and reductions in long-term institutionalisation (1,2).
Evidence from community and proactive care settings is more mixed, but suggests benefit where CGA is delivered through coordinated multidisciplinary teams with structured follow-up, particularly in improving function and supporting independence (3-7).
Maximising CGA impact:
CGA is most effective when delivered as a coordinated, continuous process. In its absence, care fragments, with individual assessments undertaken in isolation rather than contributing to a cumulative understanding over time.
Why does continuity matter?
Continuity matters because frailty is dynamic. A person’s needs change over time, often in response to illness, recovery, or changes in their social circumstances. Without continuity, care becomes episodic, with each interaction starting from a partial understanding of the individual.
Continuity enables clinicians to build a cumulative understanding of the person over time, recognising patterns, identifying change early, and adjusting care proactively. This cumulative understanding allows care to evolve rather than reset at each contact.
In this way, continuity is fundamental to Comprehensive Geriatric Assessment. CGA is not a one-off assessment, but a continuous, lifelong process. It depends on knowledge of the individual being built, shared, and carried forward across interactions and across teams.
What is the evidence for continuity?
There is a consistent body of evidence demonstrating that continuity improves outcomes. Studies have shown that when patients experience continuity of care, they are more satisfied with their care(8), receive higher quality care(9), have fewer A&E attendances and hospitalisations(10), live longer(6,11), and experience more cost-effective care(12).
In the context of frailty, where needs are complex and evolving, continuity is not optional. It enables CGA to function as a continuous process of care.
Why does coordination matter?
Continuity alone is not sufficient. Comprehensive Geriatric Assessment (CGA) also depends on coordination, with multiple professionals working together as a single team around the individual.
In practice, frailty care involves general practice, community services, hospital teams, social care, and the voluntary sector. Coordination brings these organisations and activities together, enabling shared understanding and aligned decision-making across teams.
Without it, CGA cannot function as a single model of care.
What is the evidence for coordination?
There is consistent evidence that coordinated, multidisciplinary approaches are more effective than single-domain interventions(13).
International models of integrated frailty care, including PRISMA in Canada(14), GRACE in the United States(15,16), and the Walcheren model in the Netherlands(17,18), demonstrate improved outcomes when care is delivered through structured, coordinated teams.
How is coordination different from integration?
In the NHS, integration and coordination are often used interchangeably. For the purposes of this series, it is helpful to distinguish between them.
Integration describes how services and organisations are structured. It includes:
- Partnerships between organisations
- Pooled budgets and joint commissioning
- Formation of integrated teams or systems
- Governance and strategic alignment
Integration is structural. It describes how the system is organised.
Coordination, by contrast, describes how care is delivered around the individual and how it is experienced by patients and frontline teams.
Coordination often depends on integration, but integration does not guarantee coordination. It is possible to have integrated structures where care remains fragmented.
Put simply, integration is how the system is organised. Coordination is how care is experienced.
Bringing this together
Population health management in frailty identifies need and delivers evidence-based interventions. For moderate to severe frailty, care depends on continuity, coordination, and Comprehensive Geriatric Assessment.
This raises a more fundamental question. If these are the core components of effective care, how do they come together in practice? Where does continuity sit? How is coordination achieved? And how is CGA delivered as a single, shared process across teams?
Where this series goes next
In the next article, we will explore how systems can organise around continuity, coordination, and CGA, drawing on teams already delivering elements of this and what it would take to bring them together.
The pieces are already there. The question is how to make them work as a whole.
References
- Ellis G, et al. Comprehensive geriatric assessment for older adults admitted to hospital. Cochrane Database Syst Rev. 2017 Sep 12;9(9):CD006211. doi: 10.1002/14651858.CD006211.pub3.
- Tan I. et al. Comprehensive geriatric assessment for older adults admitted to the hospital as emergencies: a meta-analysis of randomized controlled trials, Geriatric Nursing, Volume 64,
- https://doi.org/10.1016/j.gerinurse.2025.05.013.
- Stuck A., et al. Home visits to prevent nursing home admission and functional decline in elderly people: systematic review and meta-regression analysis. JAMA 2002;287(8):1022-8.
- Beswick A., et alComplex interventions to improve physical function and maintain independent living in elderly people: a systematic review and meta-analysis. Lancet 2008;371(9614):725-35.
- Briggs R, et al. Comprehensive Geriatric Assessment for community‐dwelling, high‐risk, frail, older people. Cochrane Database of Systematic Reviews 2022, Issue 5. Art. No.: CD012705. DOI: 10.1002/14651858.CD012705.pub2. Accessed 26 March 2026.
- Attwood D, et al. Does proactive care in care homes improve survival? A quality improvement project. BMJ Open Qual. 2024 Jun 4;13(2):e002771. doi: 10.1136/bmjoq-2024-002771.
- Case Study: Jean Bishop Integrated Care Centre. Community Health Partnerships 2025
- Baker R, et al. Exploration of the relationship between continuity, trust in regular doctors and patient satisfaction with consultations with family doctors. Scand J Prim Health Care. 2003 Mar;21(1):27-32. doi: 10.1080/0283430310000528.
- Romano M,. et al. The Association Between Continuity of Care and the Overuse of Medical Procedures. JAMA Intern Med. 2015 Jul;175(7):1148-54. doi: 10.1001/jamainternmed.2015.1340.
- Kohnke H, et al. Association between continuity of care in Swedish primary care and emergency services utilisation: a population-based cross-sectional study. Scand J Prim Health Care. 2017 Jun;35(2):113-119. doi: 10.1080/02813432.2017.1333303.
- Barker I, et al. Association between continuity of care in general practice and hospital admissions for ambulatory care sensitive conditions: cross sectional study of routinely collected, person level data. BMJ 2017; 356:j84 doi: https://doi.org/10.1136/bmj.j84
- Pereira Gray D., et al. Continuity of care with doctors-a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018 Jun 28;8(6):e021161. doi: 10.1136/bmjopen-2017-021161.
- Hendry A, et al. Integrated Care Models for Managing and Preventing Frailty: A Systematic Review for the European Joint Action on Frailty Prevention (ADVANTAGE JA). Transl Med UniSa. 2019 Jan 6;19:5-10. PMID: 31360661; PMCID: PMC6581495.
- Hébert, R. et al. ‘Impact of PRISMA, a coordination-type integrated service delivery system for frail older people in Quebec (Canada): a quasi-experimental study’, Journals of Gerontology Series B, 2010 65B(1), pp.107–118. DOI: 10.1093/geronb/gbp027.
- Counsell, S.R. et al. ‘Geriatric care management for low-income seniors: a randomized controlled trial’, JAMA 2007, 298(22), pp.2623–2633. DOI: 10.1001/jama.298.22.2623. [GRACE model: RCT; in-home CGA by NP/SW team; improved QoL, quality of care, reduced ED visits; cost-saving by year 2 for most complex patients]
- Counsell, S.R. et al. ‘Cost analysis of the Geriatric Resources for Assessment and Care of Elders care management intervention’, Journal of the American Geriatrics Society 2009, 57(8), pp.1420–1426. DOI: 10.1111/j.1532-5415.2009.02383.x. [GRACE cost analysis: savings emerged year 2, increased year 3]
- Looman, W.M. et al. ‘The effects of a pro-active integrated care intervention for frail community-dwelling older people: a quasi-experimental study with the GP-practice as single entry point’, BMC Geriatrics 2016, 16(1), 43. DOI: 10.1186/s12877-016-0214-5
- Looman, W.. et al. ‘Cost-effectiveness of the Walcheren Integrated Care Model intervention for community-dwelling frail elderly’, Family Practice 2016, 33(2), pp.154–160. DOI: 10.1093/fampra/cmv106.